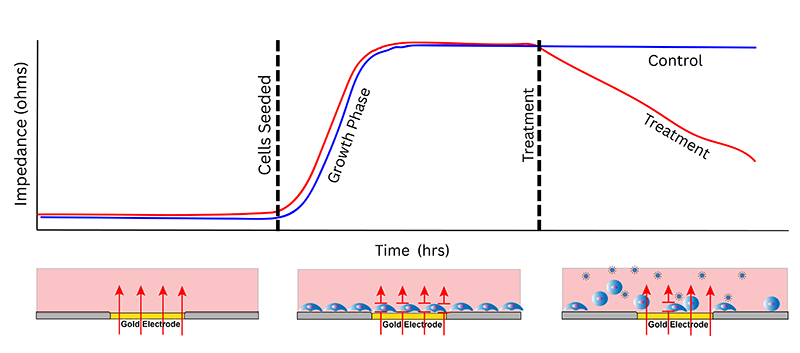
ECIS® (Electric Cell-substrate Impedance Sensing) is a real-time, label-free, impedance-based method to study cell behaviors in tissue culture including barrier function/TEER, growth/viability, migration, and more - behaviors driven by the cytoskeleton.
ECIS® uses a small non-invasive alternating current (I) that is applied across the gold-film electrode pattern at the bottom of ECIS arrays. This results in a potential (V) across the electrodes which is measured by the ECIS instrument. The cell membranes insulate the incoming current resulting in an increase in impedance. The impedance (Z) is determined by Ohm’s law Z = V/I.
When cells attach and cover the electrode, they impede current flow and increase measured impedance.
As cell morphology or junctional complexes change, the impedance of the current will also change
Time-course data allows monitoring of the dynamics of the cell behaviors from attachment to confluence to barrier formation.
Frequency of the alternating current changes how current travels around or through cell layers on gold electrodes, so different frequencies emphasize different cell behaviors. Low AC frequency causes the majority of the current to travel the resistive pathway around the cells and through the cell-cell junctions, whereas high AC frequency causes the majority of the current to flow the capacitive pathway through the cell.
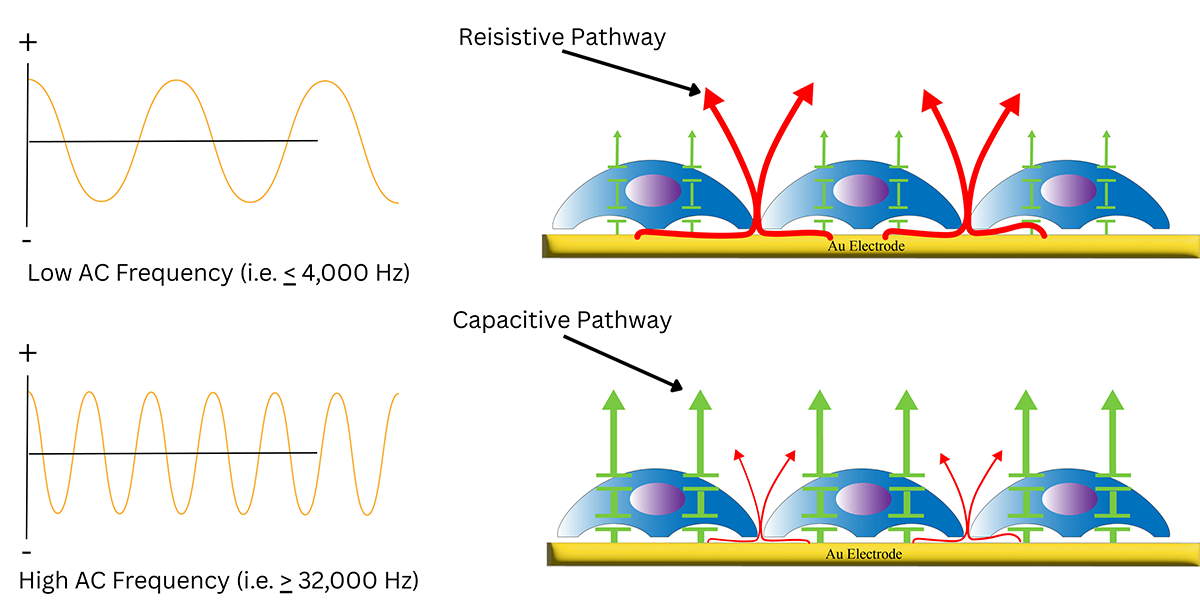
Cellular impedance contains two components-resistance and capacitance which are largely dictated by AC frequency. ECIS® measures complex impedance which isolates these two pathways so they can be viewed separately without the interfering contribution of the other pathway.
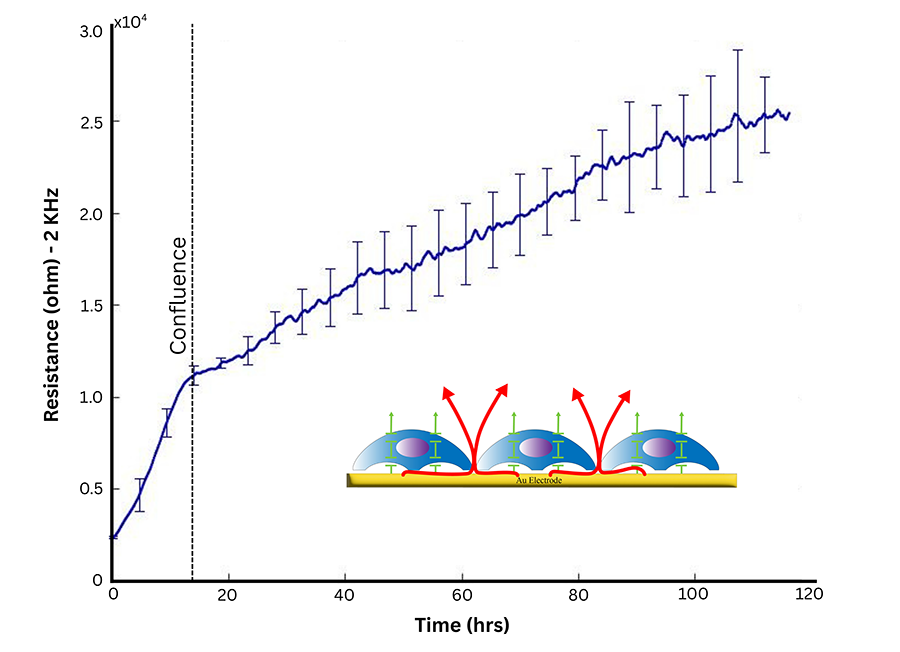
At lower frequencies, most current flows under and between adjacent cells. This makes measurements highly sensitive to cell-cell junction dynamics and attachment-related changes.
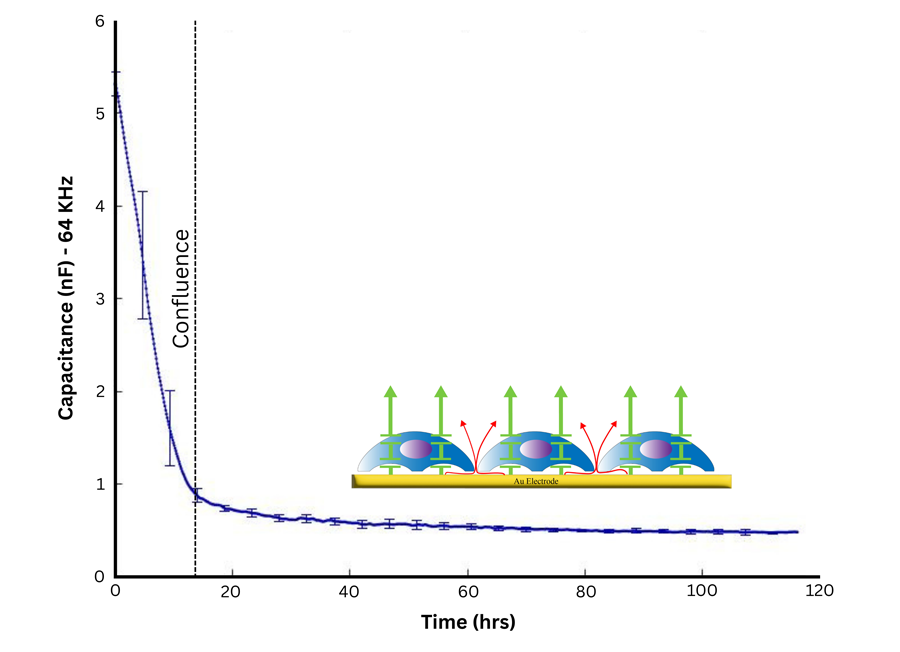
At higher frequencies, more current couples capacitively through cell membranes, making measurements more sensitive to how much the electrode is covered (useful for growth rate, viability, and migration).
© 2026 Applied BioPhysics, Inc.
185 Jordan
Road Troy, NY 12180 / Phone: 518-880-6860 / Toll Free: 866-301-ECIS (3247) / Fax: 518-880-6865