Quantitatively track cell growth from seeding to confluence using ECIS® Z-theta. Multi-frequency impedance and high-frequency capacitance provide a clearer, more direct readout of proliferation.
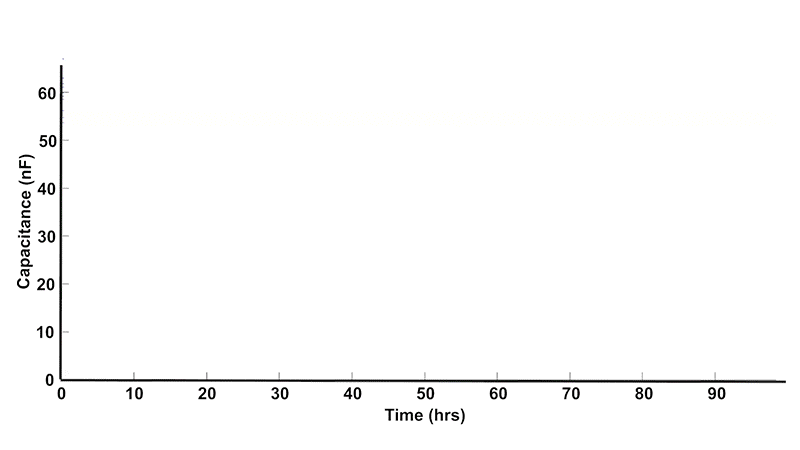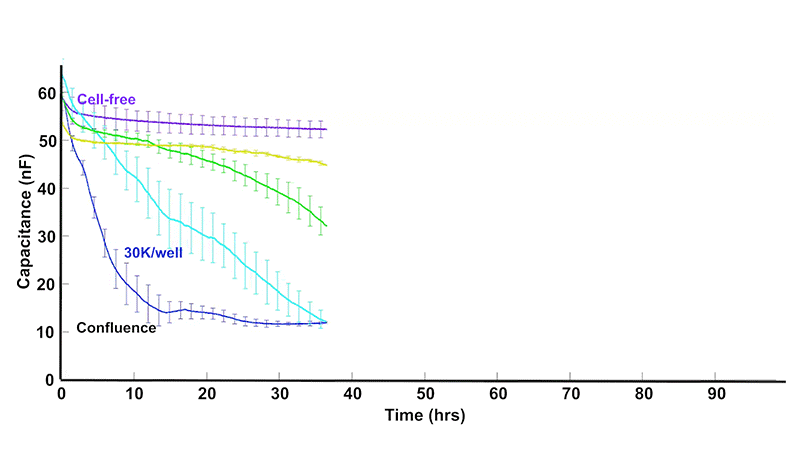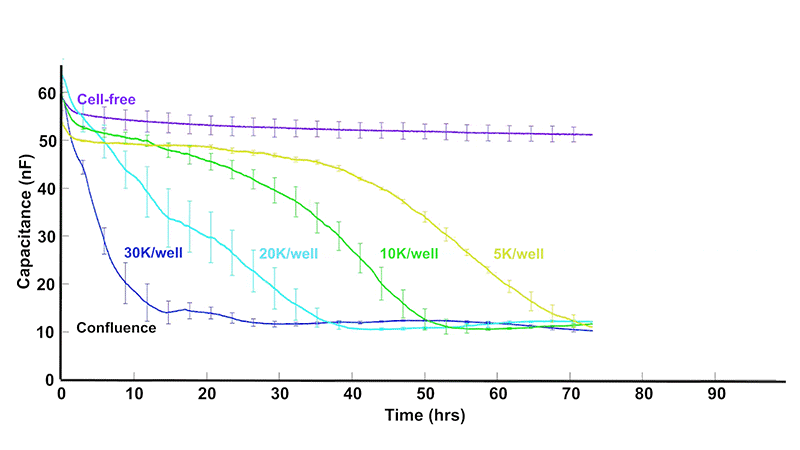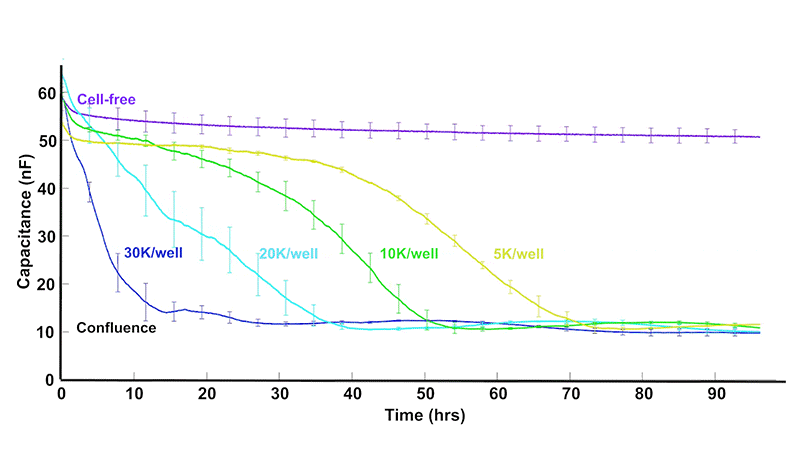
As cells proliferate, changes in cell number and morphology alter the electrical properties at the electrode surface. ECIS® captures these changes continuously, revealing growth dynamics that are difficult to observe with endpoint assays.
Observe proliferation continuously instead of relying on single time points.
Track the full progression of cell coverage across the electrode.
Quantitatively compare growth rates across treatments and environments.
Total or simple impedance measurements can be ambiguous, as multiple cellular behaviors may produce similar responses. ECIS® Z-theta resolves this by using complex impedance and separates impedance into resistance and capacitance across multiple frequencies.
At higher AC frequencies, capacitance decreases in proportion to the fraction of the electrode covered by cells, making it a highly sensitive and quantitative indicator of proliferation.
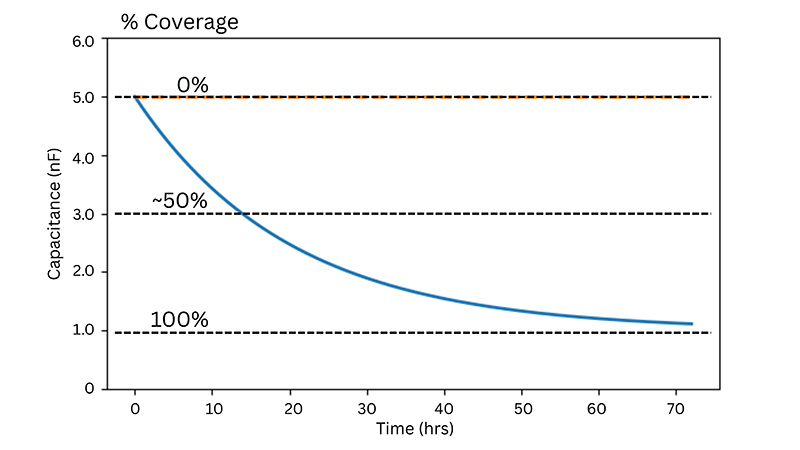
Some experimental protocols, such as cell proliferation, require sparse inoculations leading to a variance of cell density at the bottom of the well. Large electrodes (20idf Array) or a large collection of small electrodes (10E+ Array) increases the sampling size resulting in less variability.
Order Arrays Talk to an Expert
*See our publications page to explore more publications with ECIS®
© 2026 Applied BioPhysics, Inc.
185 Jordan
Road Troy, NY 12180 / Phone: 518-880-6860 / Toll Free: 866-301-ECIS (3247) / Fax: 518-880-6865