Traditional scratch assays often suffer from poor reproducibility and damage to the extracellular matrix. ECIS® replaces mechanical scratching with a precisely defined electrical wound and monitors closure in real time using impedance.
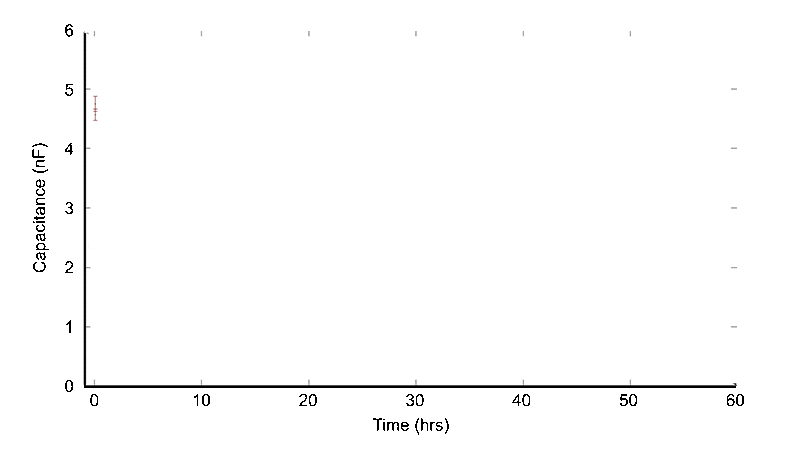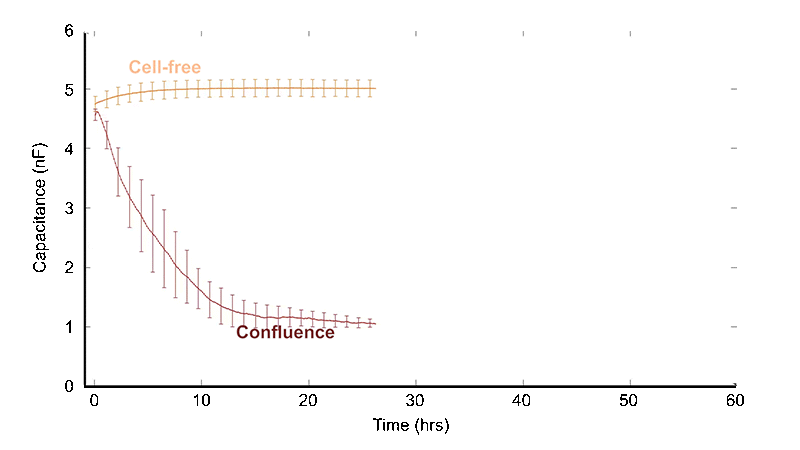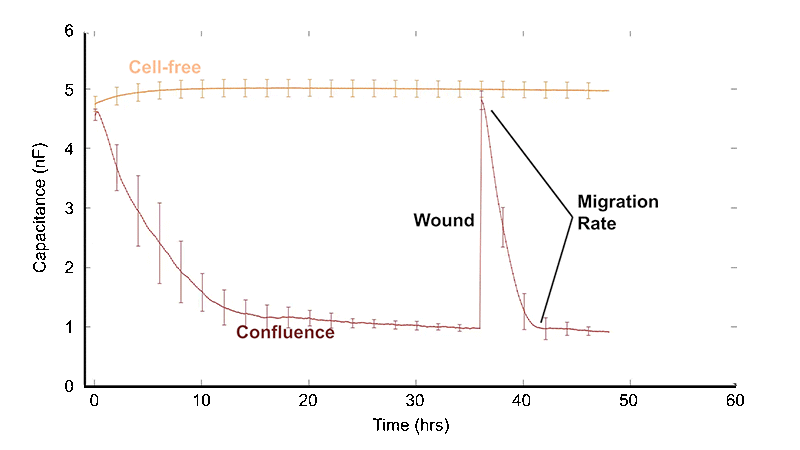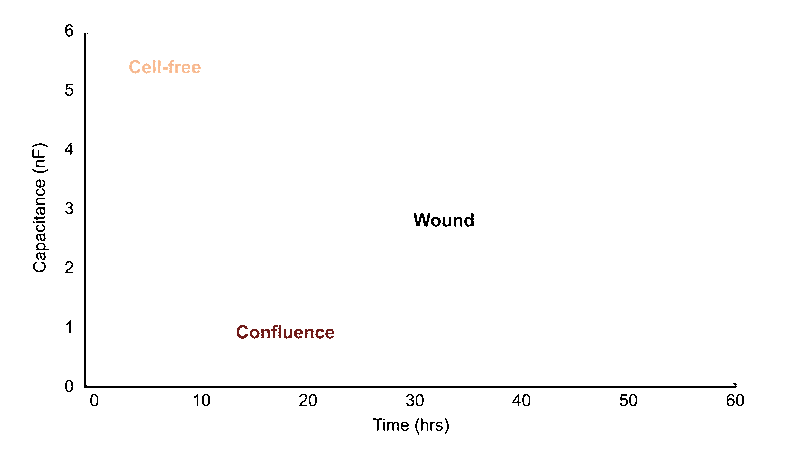
Conventional wound healing assays typically begin with a confluent monolayer that is mechanically scraped with a pipette tip or needle to create a gap — often disrupting adsorbed proteins and extracellular matrix. The gap is then inspected microscopically over hours to days as cells migrate to fill the wound.
| Traditional Scratch Assay | ECIS Wound Healing Assay | |
|---|---|---|
| Measure Cell Migration | ||
| Precise Wounding | ||
| Real-Time Data | ||
| Automated | ||
| Highly Reproducable | ||
| Matrix Preservation |
The ECIS® Wound Healing Assay replaces the traditional "scratch" assay. Instead of disrupting the cell layer mechanically to form a gap and following the migration of cells to "heal" the wound with a microscope, we employ electric signals to both wound and monitor the healing process. ECIS® electrical wounding is only directed at the small population of cells in contact with the active 250 micrometer diameter ECIS® electrode, producing a precise 250 micrometer wound that can be verified both with the ECIS® measurement and vital staining. Unlike the traditional scratch method, the ECIS® Wound Healing Assay will not affect the extracellular matrix and protein coating.

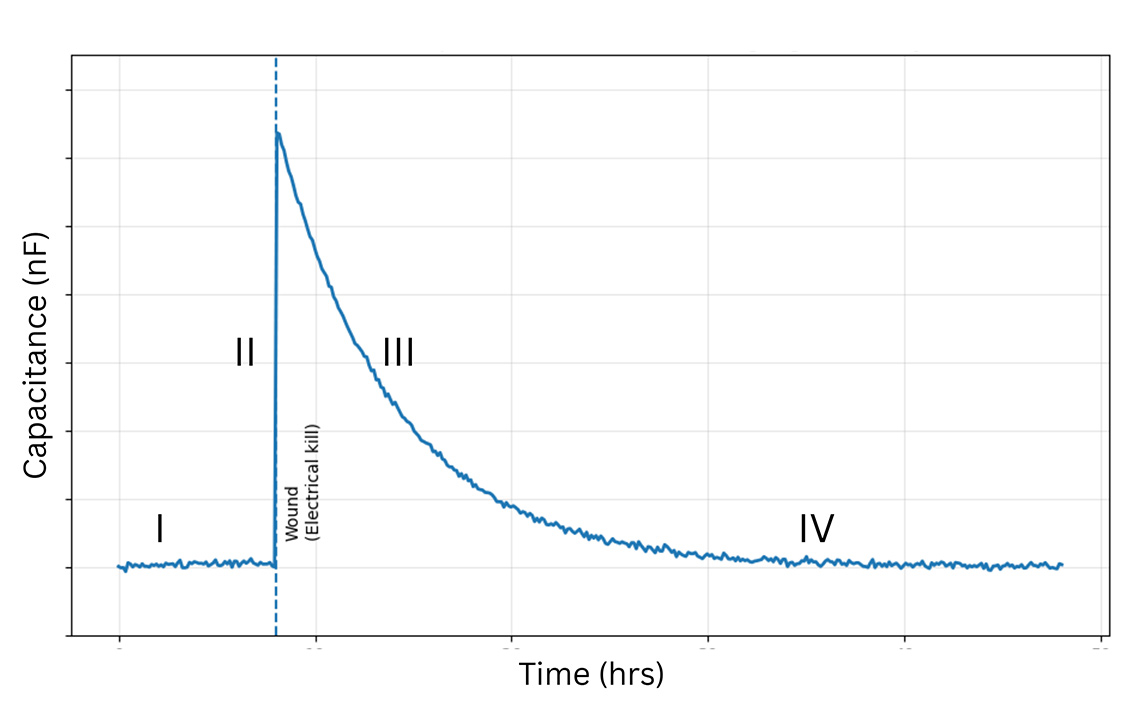
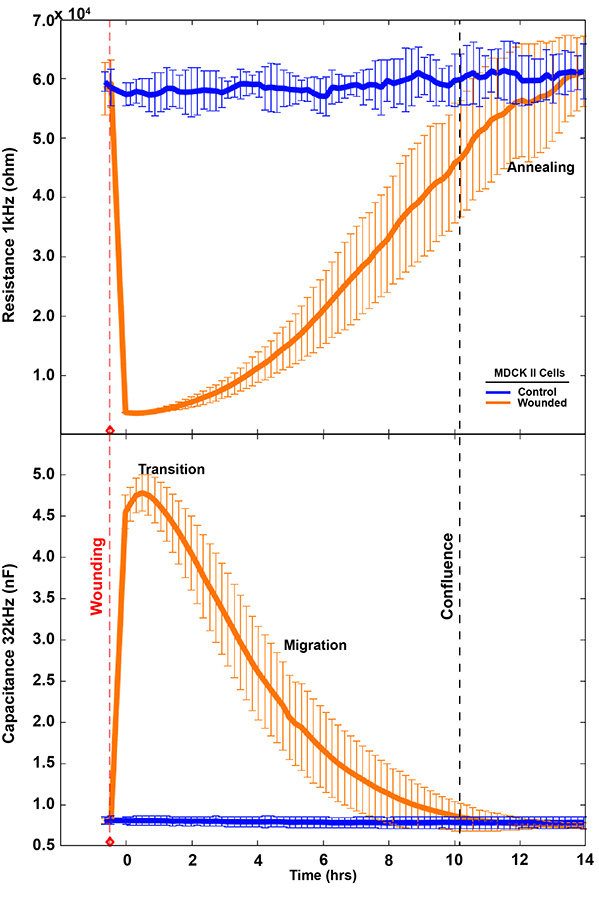
After lethal electroporation, ECIS® migration assays typically show three recovery phases: lag, fast recovery, and slow recovery. These correspond to cell transition, cell migration, and reestablishment (annealing) of cell-cell interactions.
High-frequency capacitance/impedance (>32,000 Hz) reflects electrode coverage and migration dynamics.
Low-frequency resistance (<4,000 Hz) reveals rebuilding of cell–cell connections often missed by microscopy.
Microscopy typically captures transition and migration; ECIS® adds the annealing phase.
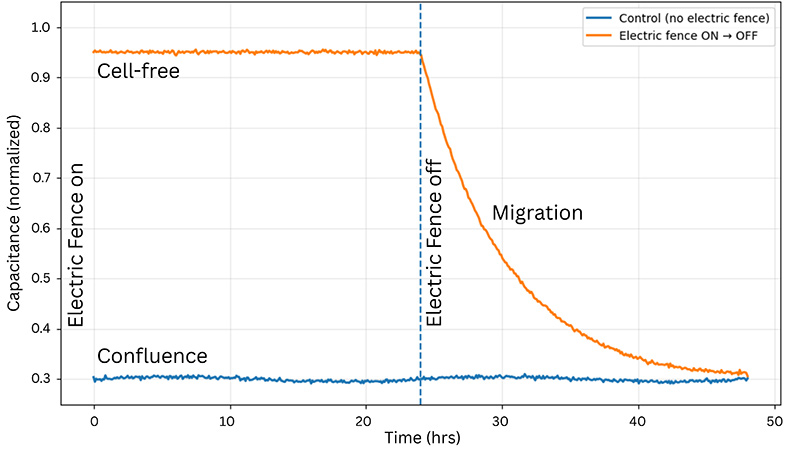
The ECIS® Electric Fence is a novel impedance-based technique to measure migration rates by preventing cells from attaching and spreading onto the measurement electrode while a confluent layer develops around it. When the fence is turned off, cells migrate into the open space left by the fence.
Using a small area of electrodes in ECIS® migration wounding assays creates a highly localized, reproducible wound by concentrating the electric field to a precise area. This improves consistency and sensitivity, enabling more accurate measurement of cell migration during wound closure. We recommend the 8w1E or 96w1E+ arrays for wound-healing migration assays.
Order Arrays Talk to an Expert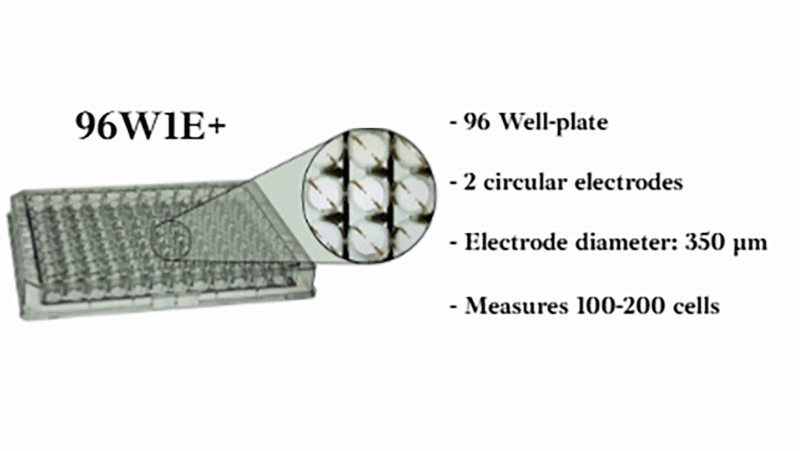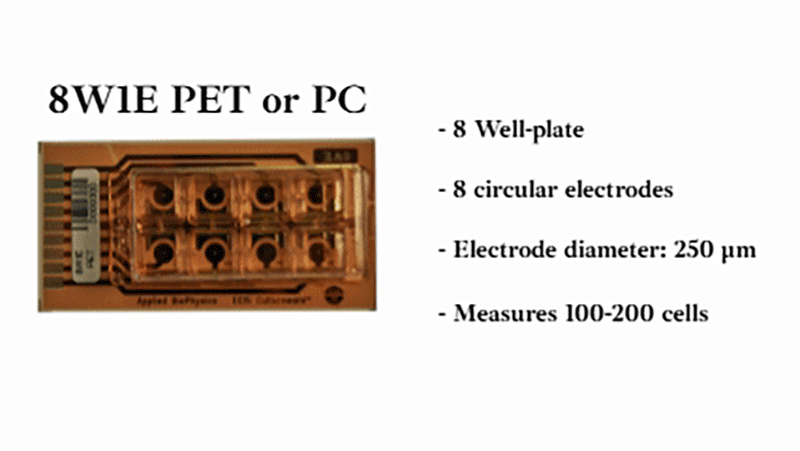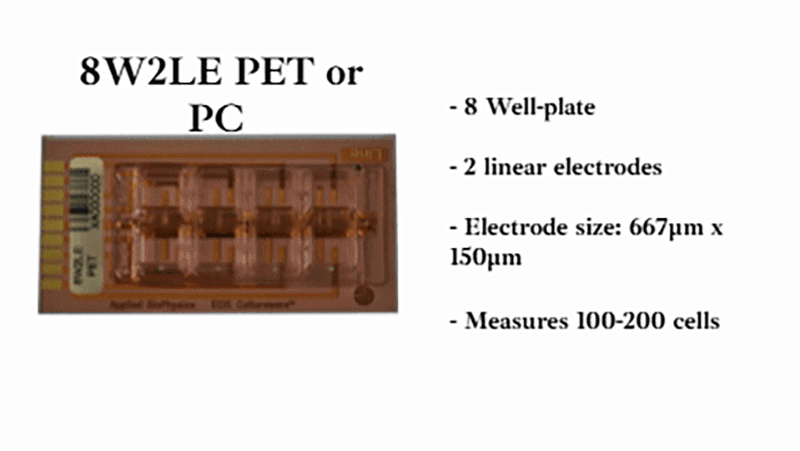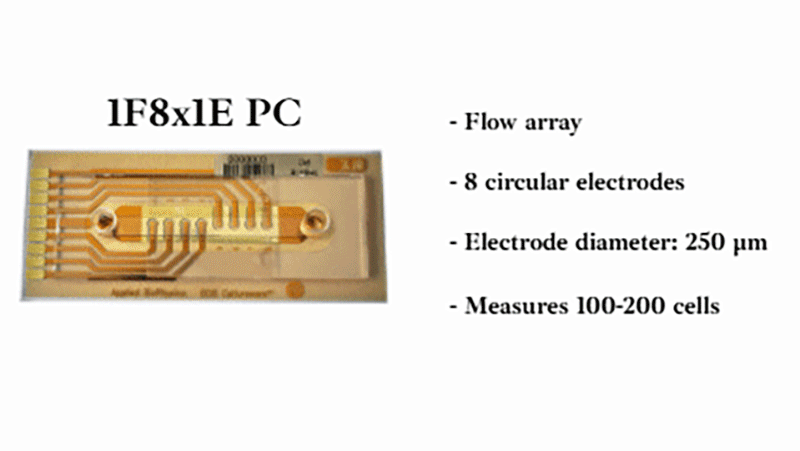
*See our publications page to explore more publications with ECIS®
© 2026 Applied BioPhysics, Inc.
185 Jordan
Road Troy, NY 12180 / Phone: 518-880-6860 / Toll Free: 866-301-ECIS (3247) / Fax: 518-880-6865