Cytotoxicity and viability can be challenging to measure with traditional qualitative assays. ECIS® enables quantitative, real-time, continuous, label-free monitoring of cytotoxic responses, while cells remain under incubated conditions.
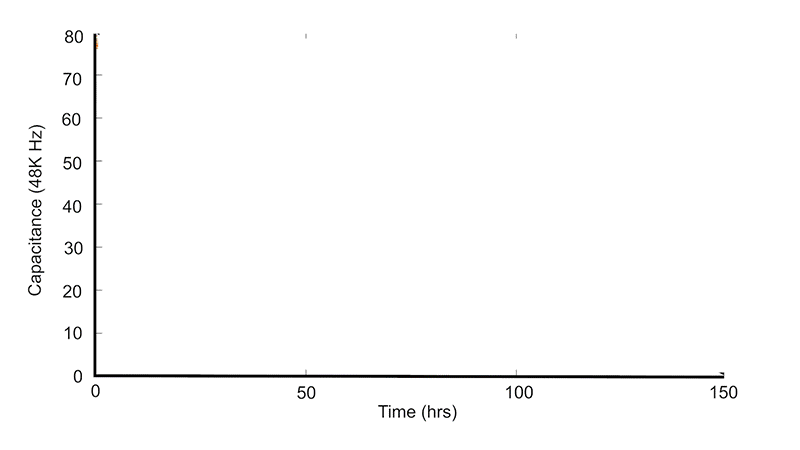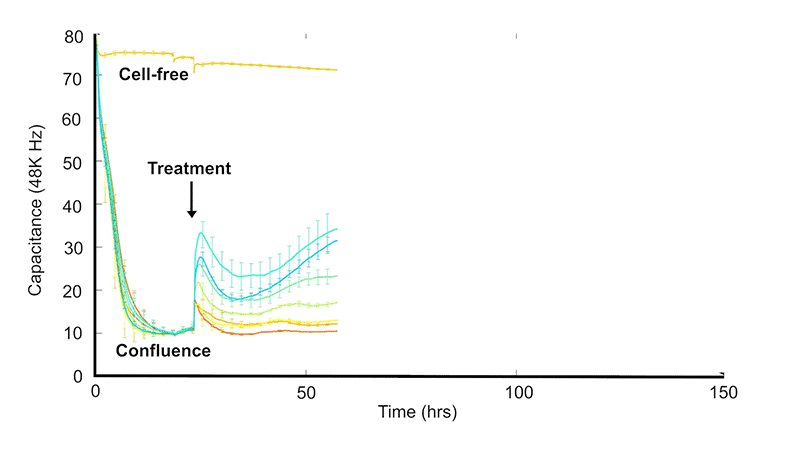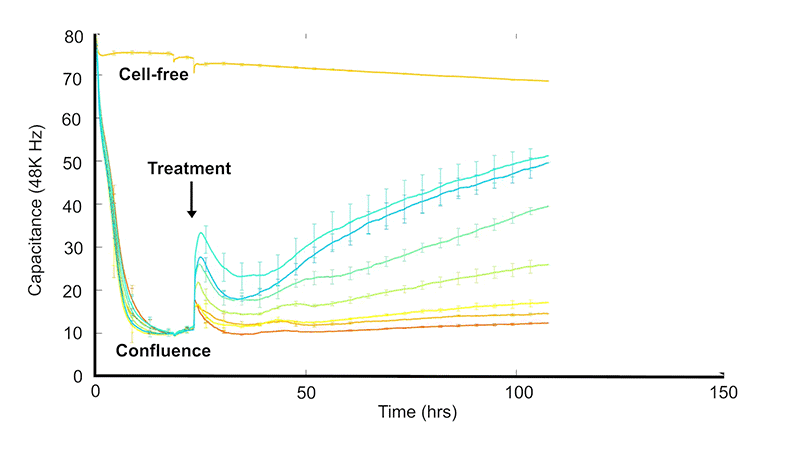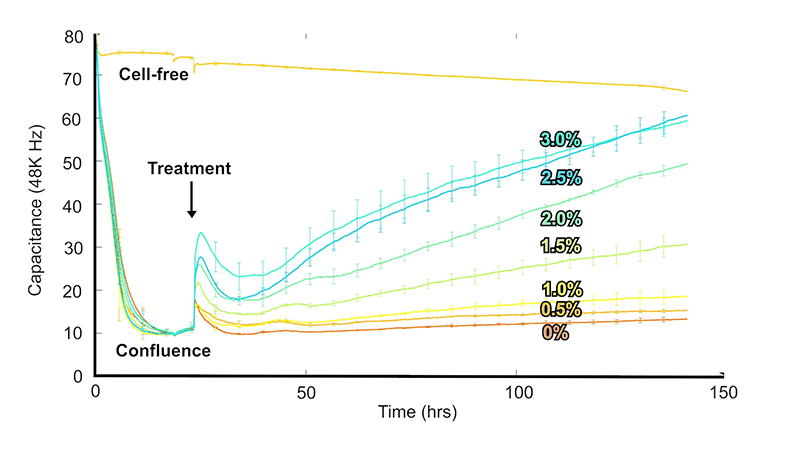
ECIS® technology measures electrical impedance changes as cells attach and spread over gold film electrodes. As cultures grow toward confluence, impedance typically rises (and capacitance falls) and plateaus. When a cytotoxic substance is introduced—such as a compound or pathogen—cells lose adhesion and detach, producing a measurable drop in impedance that tracks the cytotoxic response over time.
Capture when toxicity begins and how rapidly the response progresses.
Quantitatively compare dose, exposure time, and recovery across experimental groups.
Monitor continuously without fluorescent or colorimetric endpoints.
Standard impedance works well for many cytotoxicity assays, but the impedance subcomponent capacitance can be especially useful for measuring cell death.
At higher AC frequencies (e.g., >32,000 Hz), most current capacitively couples through cell membranes rather than taking resistive pathways around cells. This makes high-frequency capacitance ideal for cell coverage and viability assays.
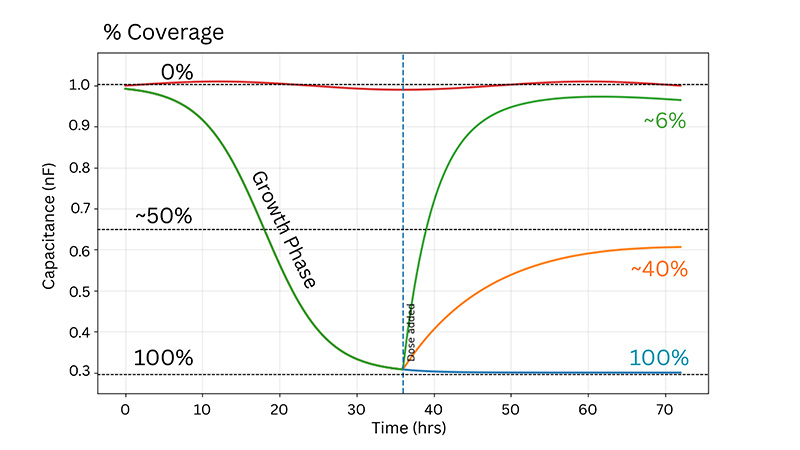
Practical interpretation:
Capacitance decreases in an approximately linear manner with electrode coverage—0% coverage for cell-free electrodes, falling to a plateau at 100% coverage or confluence—then increasing again as cells detach during cytotoxicity.
What Is ECIS?Cytotoxicity measurements often benefit from increased sampling size to reduce variability, especially with uneven cell distribution across the well.
Order Arrays Talk to an Expert
*See our publications page to explore more publications with ECIS®
© 2026 Applied BioPhysics, Inc.
185 Jordan
Road Troy, NY 12180 / Phone: 518-880-6860 / Toll Free: 866-301-ECIS (3247) / Fax: 518-880-6865