Epithelial and endothelial monolayers regulate molecular passage across tissues. ECIS® provides highly sensitive, real-time, continuous measurements of barrier resistance to support in vitro permeability and junction dynamics studies.
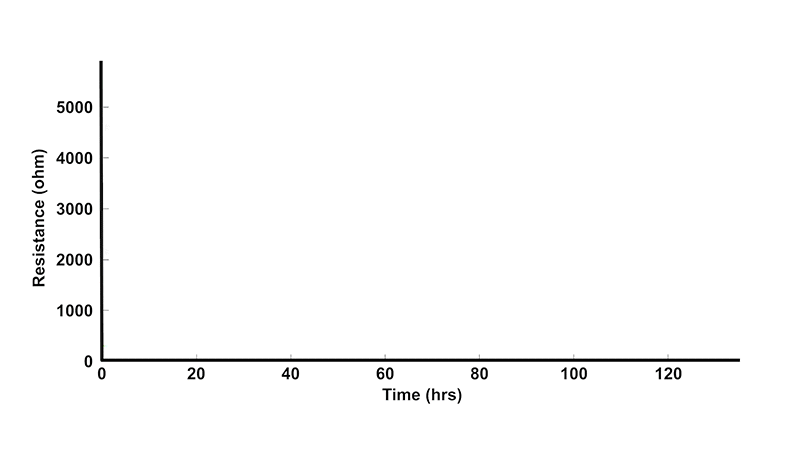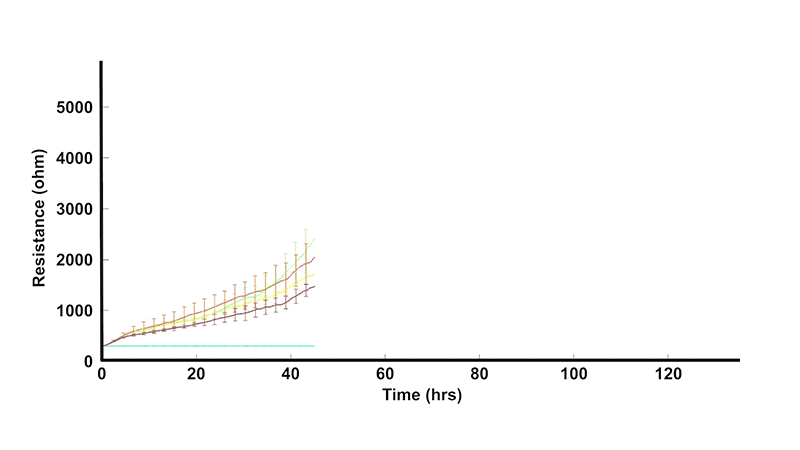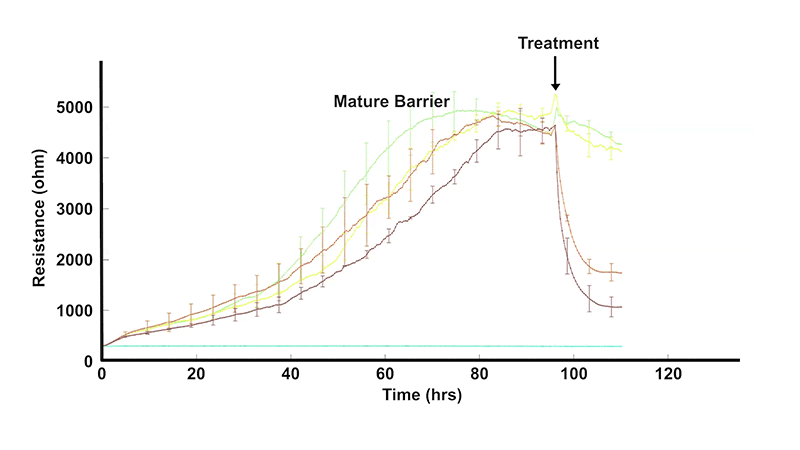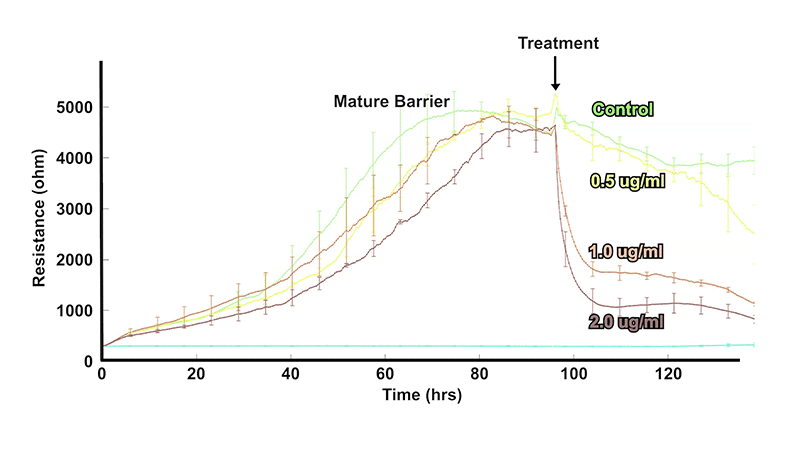
In vivo, barriers are provided by monolayers of epithelial or endothelial cells. These cell layers play a key role regulating the free movement of molecules between different tissues and/or interstitial compartments. In many diseases, as well as in inflammation, these barriers become compromised, and hence, measuring their permeability is of considerable interest to cell biologists and the health community in general.
Monitor the formation, strengthening, and disruption of cell-cell junctions continuously.
Use low-frequency resistance to track monolayer tightness as conditions change.
Quantify barrier behavior in epithelial and endothelial cultures without the need for labels.
ECIS® Z-theta measures complex impedance across multiple AC frequencies and separates impedance into series resistance (R) and capacitance (C). For barrier function studies, isolating low-frequency resistance provides a practical way to monitor cell-cell junctional barrier formation over time.
At lower frequencies, membrane impedance is high and current preferentially follows solution paths under cells and between cells—making resistance sensitive to junctional tightness.
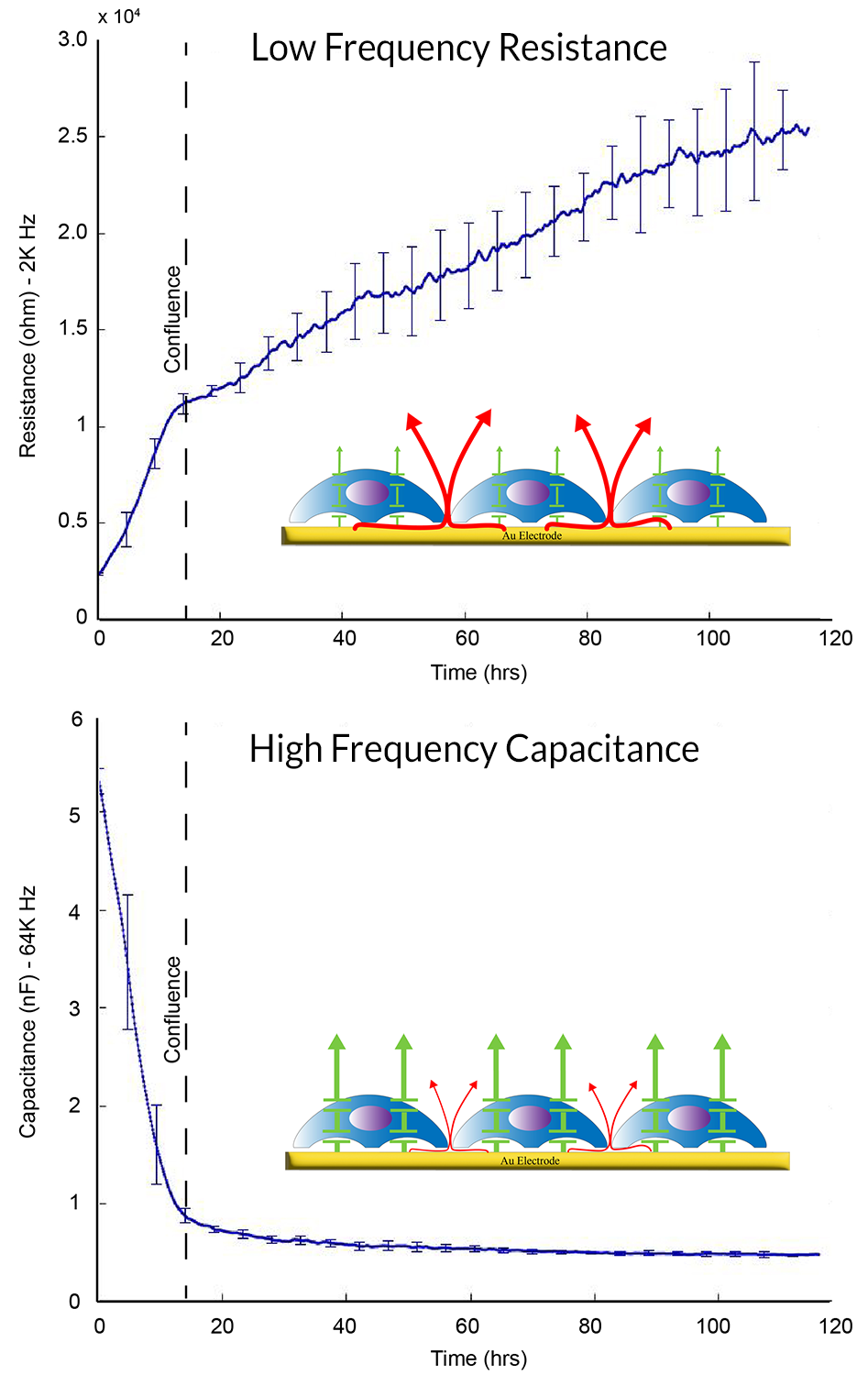
In many epithelial layers, current resistance under cells (attachment) is small relative to the paracellular path. For weaker barriers (e.g., some endothelial layers), under-cell resistance can complicate interpretation.
With the ECIS® Giaever-Keese mathematical modeling, time-course changes in barrier function resistance (Rb) and attachment under-cell resistance (alpha) can be separated and presented independently.
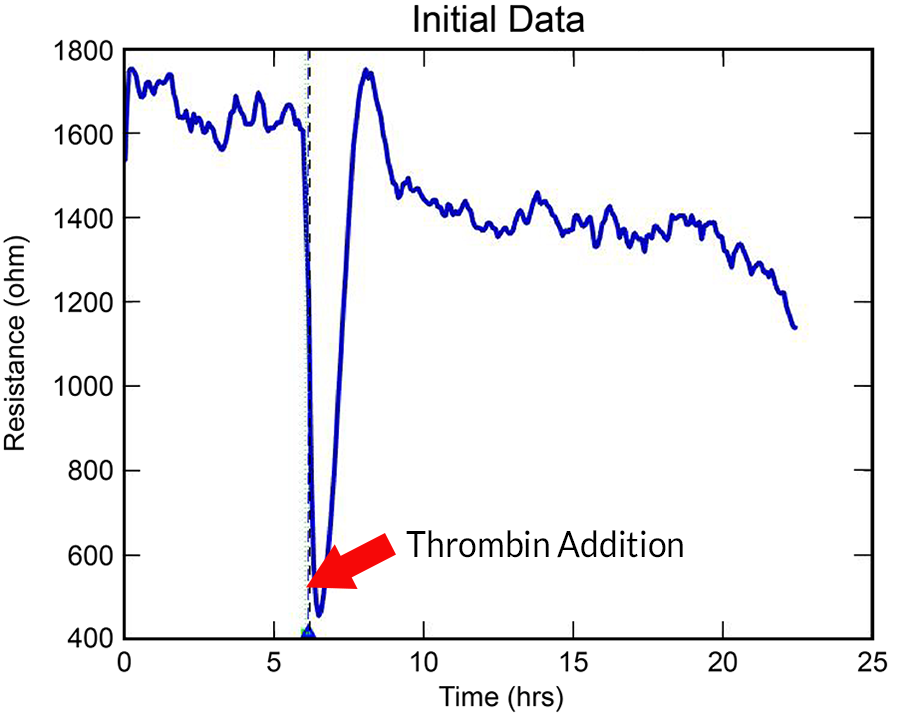
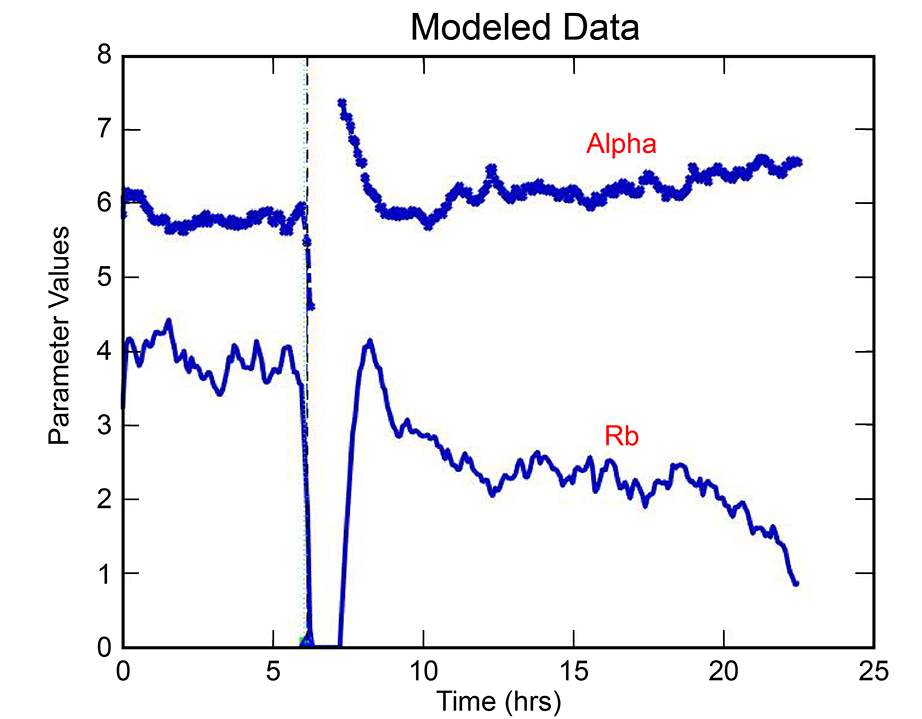
Barrier function experiments often benefit from larger electrode sampling area, especially when inoculation density varies across a well.
Order Arrays Talk to an Expert
*See our publications page to explore more publications with ECIS®
© 2026 Applied BioPhysics, Inc.
185 Jordan
Road Troy, NY 12180 / Phone: 518-880-6860 / Toll Free: 866-301-ECIS (3247) / Fax: 518-880-6865